Call Us: +27 63 169 8798
Potassium Sorbate Structure – Chemical Formula and Molecular Properties
Understanding the structure of potassium sorbate is important for industries that use it as a preservative. Potassium sorbate is widely used in food, beverage, cosmetic, and pharmaceutical products to prevent microbial growth and extend shelf life.
Potassium sorbate is the potassium salt of sorbic acid, an unsaturated fatty acid naturally found in some berries. Because of its stability and effectiveness, potassium sorbate has become one of the most common preservatives used in food manufacturing.
What Is Potassium Sorbate?
Potassium sorbate is an organic compound that functions as a food preservative and antimicrobial agent. It stops the growth of mold, yeast, and certain bacteria in food and beverage products.
The compound is typically available as a white crystalline powder or granules that dissolve easily in water. Due to its mild taste and high effectiveness, potassium sorbate is widely used in processed foods and beverages.

Potassium Sorbate Chemical Formula
The chemical formula of potassium sorbate is:
C₆H₇KO₂
This formula shows that the compound contains:
- 6 carbon atoms
- 7 hydrogen atoms
- 2 oxygen atoms
- 1 potassium atom
The molecular weight of potassium sorbate is approximately 150.22 g/mol.


Potassium Sorbate Structure

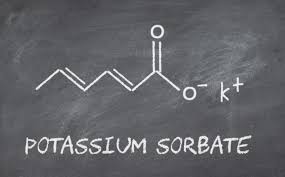
The potassium sorbate structure consists of a six-carbon chain with two double bonds and a carboxylate group bonded to a potassium ion.
The structural formula is commonly written as:
CH₃–CH=CH–CH=CH–COO⁻ K⁺
Key Components of the Structure
The molecular structure includes several important features:
Carbon Chain
The backbone of potassium sorbate contains a six-carbon chain with conjugated double bonds. These double bonds contribute to the molecule’s chemical stability.
Carboxylate Group
The molecule contains a carboxylate group (COO⁻). This functional group allows potassium sorbate to dissolve easily in water.
Potassium Ion
The negative charge of the sorbate ion is balanced by a potassium ion (K⁺), forming a stable salt.
This combination yields a stable, soluble, and effective preservative.
How the Structure Affects Its Preservative Function
The molecular structure of potassium sorbate plays a major role in its antimicrobial activity.
When potassium sorbate dissolves in water, it separates into:
- Potassium ions (K⁺)
- Sorbate ions (C₆H₇O₂⁻)
The sorbate ion interferes with microorganisms’ metabolic processes. It prevents mold and yeast from reproducing, which helps preserve food products.
This mechanism makes potassium sorbate especially effective in acidic environments, such as fruit juices
Physical and Chemical Properties
Potassium sorbate has several properties that make it ideal for use as a preservative.
Physical Properties
- Appearance: White crystalline powder
- Odor: Mild or odorless
- Solubility: Highly soluble in water
- Stability: Stable under normal storage conditions
Chemical Properties
- Chemical formula: C₆H₇KO₂
- Molecular weight: 150.22 g/mol
- pH effectiveness range: below pH 6.5
These properties allow potassium sorbate to perform reliably in many formulations.
Industrial Uses of Potassium Sorbate
Because of its chemical structure and antimicrobial activity, potassium sorbate is used in many industries.
Food and Beverage Industry
Potassium sorbate is commonly used as a food preservative in products such as:
- Fruit juices and soft drinks
- Cheese and dairy products
- Wine and alcoholic beverages
- Baked goods and pastries
- Sauces and processed foods
It prevents microbial growth while maintaining the taste and quality of food products.
Cosmetic and Personal Care Products
Potassium sorbate is also used in cosmetics and skincare products. It helps prevent contamination by microorganisms.
Typical applications include:
- Skin creams and lotions
- Liquid soaps
- Cosmetic emulsions
Pharmaceutical Applications
In pharmaceutical products, potassium sorbate acts as a preservative in liquid medications and supplements.
It helps maintain product stability during storage.
Advantages of Potassium Sorbate
There are several reasons why potassium sorbate is widely used in food preservation.
Effective Microbial Control
It effectively inhibits mold and yeast growth.
Mild Taste
Unlike many preservatives, potassium sorbate has minimal impact on flavor.
Safe and Widely Approved
Potassium sorbate is approved for use in many food products by regulatory authorities worldwide.
High Stability
The compound remains stable during processing and storage.
Related Product
If you are sourcing potassium sorbate for industrial or food applications, you can explore our product page:
🔗 Potassium Sorbate – Food Grade Preservative
https://brfsachem.com/product/potassium-sorbate/
https://brfsachem.com/product/potassium-sorbate/
This product is supplied for food processing, beverage manufacturing, and industrial applications.
Frequently Asked Questions
What is the structure of potassium sorbate?
The structure of potassium sorbate consists of a six-carbon chain with two double bonds and a carboxylate group bonded to a potassium ion.
Why is potassium sorbate effective as a preservative?
Potassium sorbate works by interfering with microbial metabolism, preventing mold and yeast from growing in food products.
Is potassium sorbate safe for food use?
Yes. Potassium sorbate is widely approved as a food preservative when used within recommended limits.
Conclusion
The potassium sorbate structure consists of a six-carbon chain with conjugated double bonds and a carboxylate group bonded to potassium. This structure gives the compound its stability and antimicrobial properties.
Because of these properties, potassium sorbate is generally used as a preservative in food, beverages, cosmetics, and pharmaceutical products. Its effectiveness and safety make it one of the most commonly used preservatives in modern manufacturing.